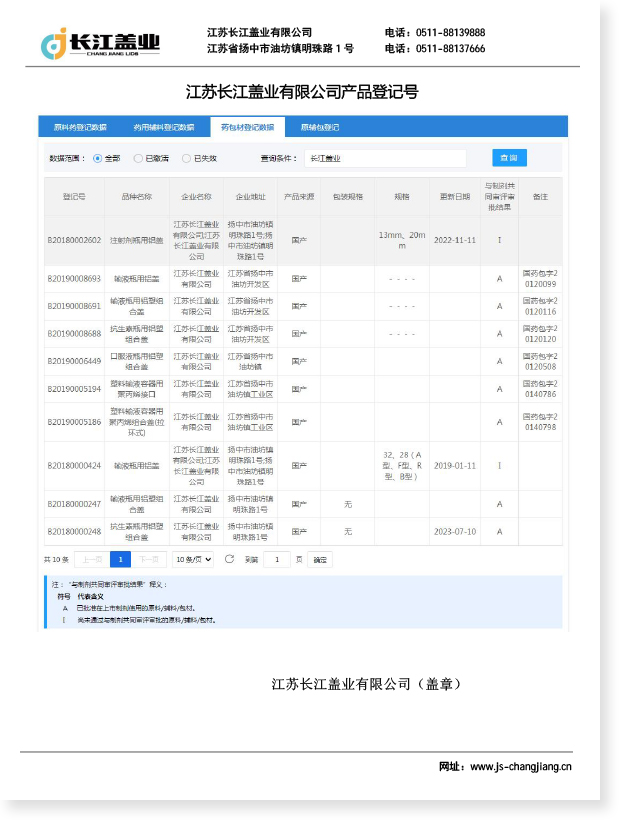
Aluminium caps for cartridge vials are precision-engineered sealing components designed to ensure the integrity, sterility, and tamper-evidence of cartridge-based pharmaceutical packaging. These caps are widely applied in insulin delivery systems, hormonal therapies, dental anesthetics, and other injectable medications that require repeated or single-dose administration via auto-injectors or pen devices. By forming a hermetic seal over the cartridge opening, aluminium caps for cartridge vials protect the drug contents from contamination, oxidation, and leakage throughout the supply chain and clinical use cycle.
As a core element of primary pharmaceutical packaging, aluminium caps for cartridge vials must comply with stringent regulatory standards including GMP guidelines and ISO 9001 quality management requirements. Jiangsu Changjiang Lids Co., Ltd., established in 2000 with over two decades of specialized manufacturing experience, produces aluminium caps with an annual capacity reaching 3 billion units, serving pharmaceutical clients globally with consistent quality and reliable performance.
Working Principle & Sealing Mechanism
The sealing function of aluminium caps for cartridge vials relies on a crimping mechanism. During the capping process, the aluminium shell is mechanically crimped around the neck finish of the cartridge, compressing an inner rubber stopper or gasket to create an airtight and sterile barrier. This process involves three key stages:
- Placement: The cap is positioned over the cartridge opening, aligning with the rubber stopper beneath.
- Crimping: A crimping tool applies radial pressure to fold the aluminium skirt tightly under the cartridge neck flange, locking the stopper in place.
- Verification: The sealed assembly is inspected for cap integrity, residual seal force (RSF), and visual defects to ensure compliance with pharmacopoeial standards.
This mechanical interlock ensures that the contents remain sterile and protected under varying temperature and pressure conditions during transportation and storage. The malleability of aluminium allows uniform deformation without cracking, making it the preferred material for pharmaceutical sealing applications.
Product Specifications & Key Parameters
Understanding the technical parameters of aluminium caps for cartridge vials helps procurement professionals and quality engineers select the right product for their specific application. The table below outlines common specifications:
| Parameter | Typical Range / Value | Significance |
|---|---|---|
| Cap Diameter | 13 mm / 20 mm (standard) | Matches cartridge neck finish dimensions |
| Material | Aluminium alloy (1xxx / 8xxx series) | Ensures malleability and corrosion resistance |
| Thickness | 0.20 – 0.35 mm | Balances seal strength and ease of crimping |
| Surface Treatment | Lacquered / anodized / plain | Corrosion protection and aesthetic customization |
| Residual Seal Force (RSF) | ≥ 25 N (typical minimum) | Indicator of effective seal integrity |
| Regulatory Compliance | USP, EP, ChP, ISO 9001, GMP | Required for global pharmaceutical market access |
| Color Options | Customizable (standard silver, gold, red, blue, etc.) | Enables product differentiation and brand identity |
| Cleanliness Level | C+A cleanroom production | Minimizes particulate contamination risk |
Application Scenarios
Aluminium caps for cartridge vials are found across a wide range of therapeutic areas where precise dosing and product sterility are critical:
- Insulin Delivery: 3 mL cartridges used in insulin pens require caps with consistent RSF values to maintain stopper integrity through repeated needle penetrations.
- Hormone Therapies (e.g., Growth Hormone, GLP-1 agonists): Biologics demand high-purity aluminium caps with minimal extractable and leachable (E&L) profiles to avoid drug interaction.
- Dental Anesthetics: Local anesthetic cartridges require tamper-evident seals and compatibility with carpule syringe systems.
- Veterinary Pharmaceuticals: Multi-dose cartridge formats for animal health products rely on aluminium caps to prevent cross-contamination between administrations.
- Combination Devices (Auto-Injectors / Pen Systems): Device manufacturers specify precise cap dimensions and hardness tolerances to ensure automated assembly compatibility.
Changjiang Lids supplies aluminium caps to pharmaceutical manufacturers across these segments, with product customization available in diameter, lacquer color, and surface finish to meet diverse OEM requirements.
Aluminium Caps vs. Aluminium-Plastic Combination Caps: A Detailed Comparison
Pharmaceutical cartridge sealing solutions include both pure aluminium caps and aluminium-plastic (flip-off) combination caps. Selecting the correct type depends on the device system, sterilization method, and end-user requirements. The comparison below clarifies the key differences:
| Feature | Pure Aluminium Cap | Aluminium-Plastic Combination Cap |
|---|---|---|
| Structure | Single aluminium shell | Aluminium shell + polypropylene flip-off button |
| Access Method | Needle penetration through centre disc | Plastic button removed to expose stopper |
| Tamper Evidence | Visual deformation after cap removal | High – intact flip-off confirms no prior access |
| Compatibility | Pen injectors, auto-injectors | Vials requiring manual access before injection |
| Customization | Color lacquer, embossing | Plastic button color + aluminium finish |
| Primary Use Case | Cartridge vials, pen systems | Injection vials, antibiotic vials |
| Cost | Lower per unit | Slightly higher due to dual-material construction |
Changjiang Lids manufactures both product types and can advise on the most suitable configuration based on your cartridge format, device system, and regulatory market requirements.
Quality Standards & Regulatory Compliance
Pharmaceutical packaging components are subject to rigorous international regulations. Aluminium caps for cartridge vials must meet the following key standards:
| Standard / Guideline | Issuing Body | Key Requirements |
|---|---|---|
| USP <660> / <661> | United States Pharmacopeia | Container and closure material compatibility |
| EP 3.2.1 / 3.2.9 | European Pharmacopoeia | Metal containers for pharmaceutical use |
| ISO 9001:2015 | ISO | Quality management system certification |
| GMP (21 CFR Part 211 / EU Annex 1) | FDA / EMA | Manufacturing environment and process controls |
| YBB Standards (China) | NMPA (China) | National pharmaceutical packaging material standards |
Jiangsu Changjiang Lids Co., Ltd. integrates GMP and ISO 9001 across the entire production lifecycle. The company has designed and constructed a C+A level cleanroom to minimize particulate and microbial contamination risks, supporting clients in achieving and maintaining their own regulatory approvals worldwide.
Quality Control & Inspection Checkpoints
Effective quality control for aluminium caps for cartridge vials covers both incoming material inspection and finished product verification. The following inspection parameters are routinely assessed:
Dimensional Inspection
- Cap outer diameter, inner diameter, and height measured against drawing tolerances using calibrated gauges.
- Skirt length and curl radius verified to ensure consistent crimping performance on filling lines.
Mechanical Performance Testing
- Residual Seal Force (RSF): Measures the compressive force retained on the stopper after crimping; a critical indicator of container closure integrity (CCI).
- Removal Torque: Evaluated for aluminium-plastic caps to ensure consistent user-accessible opening force.
- Leak Test: Assembled cartridge-cap units tested for helium leak or dye ingress to confirm hermetic sealing.
Visual & Surface Inspection
- Automated vision systems detect surface defects including scratches, dents, coating inconsistencies, and edge deformation.
- Lacquer adhesion and uniformity tested per applicable pharmacopoeial methods.
Chemical Compatibility Testing
- Extractables and leachables (E&L) profiling conducted in accordance with ICH Q3E guidelines.
- Heavy metal content verified to meet pharmacopoeial limits.
Storage, Handling & Maintenance Guidelines
Proper handling and storage of aluminium caps for cartridge vials prior to use is critical to maintaining their functional performance:
- Storage Conditions: Store in a clean, dry environment at room temperature (15–30°C), away from direct sunlight, moisture, and corrosive chemicals. Relative humidity should be controlled below 60% to prevent surface oxidation.
- Packaging Integrity: Keep original inner packaging sealed until immediate use. Avoid opening bags in open environments with high particulate counts.
- FIFO Inventory Management: Use a first-in, first-out rotation to avoid extended storage beyond the recommended shelf life specified on the Certificate of Analysis (CoA).
- Handling in Cleanrooms: Personnel handling caps in C or A-grade cleanroom environments must follow gowning protocols to prevent introduction of contaminants onto cap surfaces.
- Crimping Equipment Maintenance: Regularly calibrate and inspect crimping heads to ensure consistent application of sealing force. Worn tooling can result in under-crimped or deformed caps and compromised RSF values.
- Rejection of Damaged Units: Any cap showing visible deformation, surface corrosion, or incomplete lacquering should be quarantined and not used in production.
Why Choose Changjiang Lids for Aluminium Caps for Cartridge Vials
Sourcing decisions for primary pharmaceutical packaging involve balancing quality consistency, regulatory compliance, supply reliability, and cost. Changjiang Lids offers the following competitive advantages:
| Advantage | Details |
|---|---|
| Over 24 years of manufacturing experience | Founded in 2000; deep expertise in pharmaceutical packaging |
| High production capacity | 3 billion aluminium and aluminium-plastic caps annually |
| C+A level cleanroom environment | Minimizes contamination; meets EU GMP Annex 1 standards |
| ISO 9001 & GMP certified quality system | Full-process quality control from raw material to dispatch |
| Customization capability | Diameter, color, surface finish, and embossing options available |
| Yangtze River Delta location | Logistics efficiency for domestic and export supply chains |
Frequently Asked Questions (FAQ)
Q1: What is the difference between a cartridge vial cap and a standard injection vial cap?
Cartridge vials are cylindrical containers typically used in pen injectors and auto-injectors, sealed at one end with an aluminium cap and at the other with a piston stopper. Standard injection vials are sealed entirely at the top with an aluminium cap over a rubber stopper. The cap geometry, diameter tolerances, and crimping parameters differ between the two formats to accommodate their respective device interfaces.
Q2: Can aluminium caps for cartridge vials be customized with company logos or specific colors?
Yes. Aluminium caps can be produced with custom lacquer colors and, in some configurations, embossed text or logos. Color differentiation is commonly used by pharmaceutical manufacturers to distinguish dosage strengths, product lines, or market regions. Changjiang Lids offers customization options and can provide color samples and tooling development for OEM clients.
Q3: How is container closure integrity (CCI) verified for crimped aluminium caps?
CCI can be evaluated through several methods: residual seal force (RSF) measurement, helium leak testing, dye ingress testing, and headspace gas analysis. The choice of method depends on the drug product type, regulatory jurisdiction, and device configuration. RSF testing is widely used as a process control parameter during filling line qualification, while helium leak testing offers high sensitivity for sterile products.
Q4: What cleanroom grade is required for manufacturing aluminium caps for pharmaceutical use?
Pharmaceutical packaging components are typically manufactured in controlled environments. EU GMP Annex 1 and relevant national regulations specify cleanroom grades based on product contact and process exposure. Changjiang Lids has invested in a C+A level cleanroom to ensure that caps are produced and packaged with minimal particulate and microbial contamination, directly supporting clients' sterility assurance strategies.
Q5: What minimum order quantities (MOQ) are typical for aluminium caps for cartridge vials?
MOQ varies by product specification, customization requirements, and supplier capacity. For standard configurations, MOQs are generally aligned with bulk packaging units (e.g., per bag or per carton quantity). For custom colors or embossed designs, tooling lead times and minimum batch sizes apply. Clients are advised to contact Changjiang Lids directly to discuss volume requirements and confirm current lead times.
Q6: Are aluminium caps for cartridge vials compatible with automated filling and capping lines?
Yes. Aluminium caps are designed to be compatible with standard automated pharmaceutical filling and crimping equipment. Dimensional consistency across production batches is critical for reliable automated handling. Changjiang Lids maintains tight dimensional tolerances through its GMP-compliant manufacturing process and performs outgoing inspection to ensure batch-to-batch consistency.
Q7: How should aluminium caps be disposed of after use?
Used aluminium caps from pharmaceutical cartridges should be disposed of according to local medical waste regulations. Aluminium is fully recyclable; however, caps that have been in contact with drug products must follow pharmaceutical waste disposal guidelines in the relevant jurisdiction before recycling can be considered. End-users and healthcare providers should consult local regulatory guidance on sharps and pharmaceutical packaging waste management.

 English
English русский
русский