Aluminium caps for infusion bottles are primary pharmaceutical packaging components designed to seal large-volume parenteral (LVP) containers — including glass and plastic infusion bottles used for IV solutions, saline, glucose, and other critical intravenous medications. These caps serve as the final barrier between the sterile drug product and the external environment, making their performance directly linked to patient safety.
Structurally, an aluminium cap for infusion bottles typically combines a precisely formed aluminium shell with a bromobutyl or chlorobutyl rubber stopper insert. The aluminium component provides mechanical crimping force and tamper-evidence, while the rubber stopper maintains container closure integrity (CCI) and enables needle penetration for drug administration or reconstitution. Some configurations incorporate a tear-off or flip-off polypropylene disc for ease of access and visible tamper indication.
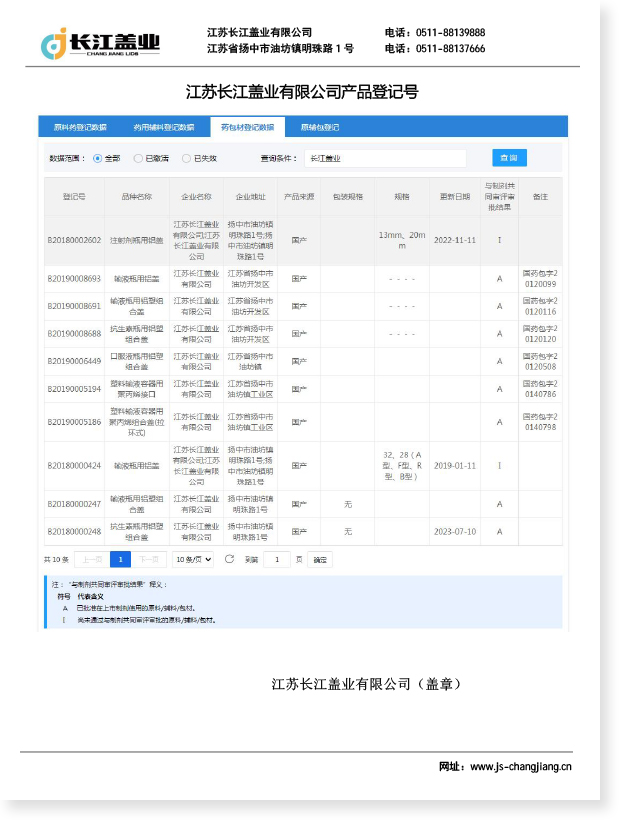
At Jiangsu Changjiang Lids Co., Ltd., aluminium caps for infusion bottles are manufactured under GMP-compliant conditions in a certified C+A level cleanroom, with an annual production capacity of 3 billion aluminium and aluminium-plastic caps. Every cap undergoes process control across the full production cycle to ensure consistent dimensional accuracy, seal integrity, and compliance with pharmaceutical regulatory standards.
Core Functions and Performance Requirements
The functional demands placed on aluminium caps for infusion bottles are stringent. Each cap must reliably perform across multiple critical parameters throughout the product's shelf life and under varied handling and storage conditions:
- Container Closure Integrity (CCI): Prevent microbial ingress, moisture transmission, and gas exchange to maintain sterility.
- Tamper Evidence: Provide visible indication of any unauthorized access or compromise before use.
- Chemical Compatibility: Aluminium and rubber components must not interact adversely with the enclosed drug product or leach harmful extractables.
- Mechanical Crimping Performance: The aluminium shell must deform uniformly during capping to achieve a consistent, leak-free seal without cracking or delamination.
- Dimensional Precision: Caps must conform to bottle finish specifications (diameter, height, skirt thickness) to ensure compatibility with automated filling and capping lines.
- Resealability: After needle penetration, the rubber insert must reseal to prevent secondary contamination.
Product Types and Configuration Overview
Aluminium caps for infusion bottles are available in several structural configurations, each suited to specific bottle types, filling line equipment, and market requirements. The table below summarizes the primary variants:
| Configuration Type | Structure Description | Typical Application | Key Feature |
|---|---|---|---|
| Plain Aluminium Cap | Single aluminium shell with rubber stopper | Standard IV infusion glass bottles | Cost-effective, high compatibility |
| Aluminium-Plastic Combination Cap (Flip-off) | Aluminium shell + PP flip-off button | Infusion bottles requiring visible tamper indication | Clear tamper evidence, easy cap removal |
| Tear-Off Aluminium Cap | Scored aluminium ring enabling manual removal | Hospital and clinical settings | Tool-free removal, fast administration |
| Coloured Aluminium Cap | Anodised or coated aluminium in specified colours | Multi-product differentiation and brand identification | Visual coding for medication error prevention |
| Large-Diameter Infusion Cap (≥32 mm) | Wide-mouth aluminium shell with oversized stopper | 500 mL / 1000 mL large-volume infusion bottles | Designed for high-volume LVP containers |
Changjiang Lids supplies aluminium caps for infusion bottles across this full range of configurations, with customisation available for dimensions, colour coding, and surface finish to meet individual client specifications.
How Aluminium Caps for Infusion Bottles Work: Sealing Mechanism
The sealing process for aluminium caps for infusion bottles involves two sequential mechanisms working in combination:
1. Crimping Seal
During the capping operation on a pharmaceutical filling line, a crimping head applies radial and downward pressure to the aluminium shell. This deforms the skirt of the cap around the bottle neck's groove or bead, creating a mechanical lock. The clamping force compresses the rubber stopper against the bottle mouth, generating a hermetic seal. Crimping force, angle, and head geometry are calibrated to ensure consistent deformation without material fracture.
2. Elastomeric Stopper Seal
The bromobutyl or chlorobutyl rubber stopper, held under constant compression by the crimped aluminium shell, provides the primary barrier against microbial contamination and moisture ingress. The elastomer also self-seals after needle withdrawal, maintaining closure integrity during multi-dose use scenarios. Stopper formulation is selected based on drug compatibility requirements — bromobutyl is preferred for moisture-sensitive products, while chlorobutyl offers advantages for certain aqueous formulations.
3. Flip-off / Tamper-Evidence Layer
In aluminium-plastic combination caps, a polypropylene disc or button is attached to the top of the aluminium shell. This component snaps off or flips open before needle insertion, providing a clear visual indicator that the cap has not been accessed. The PP element does not contribute to the primary seal but is an important safety and regulatory compliance feature in many markets.
Material Specifications and Quality Standards
The material quality of aluminium caps for infusion bottles directly affects seal performance, extractables profile, and downstream compatibility with sterilisation processes. The table below outlines key material and quality parameters:
| Parameter | Specification / Standard | Relevance |
|---|---|---|
| Aluminium alloy grade | 1xxx or 8xxx series (pharmaceutical grade) | Formability, corrosion resistance |
| Aluminium thickness | 0.18 mm – 0.25 mm (typical) | Crimping performance, skirt integrity |
| Rubber stopper type | Bromobutyl / Chlorobutyl | Drug compatibility, moisture barrier, resealability |
| Cleanroom production grade | C+A level (ISO equivalent) | Particulate and microbial control |
| Quality system | GMP + ISO 9001 | Process consistency, traceability |
| Sterilisation compatibility | Autoclave / gamma-irradiation ready (application-dependent) | Pre-sterilisation capability for aseptic filling |
| Dimensional tolerances | Per ChP / USP / EP packaging monographs | Filling line compatibility, CCI reliability |
Aluminium Cap vs. Polypropylene Cap for Infusion Bottles: Comparison
Both aluminium caps and polypropylene (PP) caps are used for infusion bottle sealing, but they serve different applications and offer distinct performance profiles. Understanding the differences helps pharmaceutical manufacturers select the appropriate closure system.
| Attribute | Aluminium Cap (with rubber stopper) | Polypropylene Cap |
|---|---|---|
| Primary container type | Glass infusion bottles | Plastic (PP/PE) infusion containers |
| Sealing method | Crimping + elastomeric compression | Snap-fit / threaded / welded |
| Needle penetration | Yes (via rubber stopper) | Varies by design (spike port) |
| Tamper evidence | Mechanical crimping + optional flip-off | Breakable ring / tamper band |
| Autoclave compatibility | High (aluminium is heat-stable) | Moderate (grade-dependent) |
| Typical usage in China market | Dominant in glass bottle LVP | Growing use in plastic LVP bags/bottles |
| Recyclability | Aluminium shell fully recyclable | PP is recyclable where facilities exist |
Changjiang Lids manufactures both aluminium caps for infusion bottles and polypropylene caps for infusion containers, enabling clients to source complementary closure solutions from a single, GMP-certified supplier.
Application Scenarios
Aluminium caps for infusion bottles are applied across a broad range of pharmaceutical production and clinical contexts:
- Large-Volume Parenterals (LVP): 250 mL, 500 mL, and 1000 mL glass bottles filled with isotonic saline, glucose solutions, Ringer's lactate, and other IV fluids. These represent the largest volume application globally.
- Antibiotic and Pharmaceutical Additions: Infusion bottles used in hospital pharmacy compounding, where a secondary drug is added via needle injection through the rubber stopper. Resealability and particulate-free inner surfaces are critical here.
- Blood Volume Expanders: Dextran, hydroxyethyl starch, and albumin infusion products, where chemical compatibility between stopper and formulation must be validated.
- Parenteral Nutrition (PN): Lipid emulsion and amino acid solutions packaged in glass infusion bottles, requiring high barrier performance and compatibility with lipid-based formulations.
- Veterinary Infusion Products: Large-format infusion solutions for animal healthcare, following similar sealing requirements to human pharmaceutical products.
Key Selection Criteria for Buyers and Procurement Teams
When evaluating suppliers of aluminium caps for infusion bottles, pharmaceutical manufacturers and procurement professionals should assess the following factors:
| Evaluation Area | Key Questions to Ask | Changjiang Lids Position |
|---|---|---|
| Regulatory compliance | Is the facility GMP-certified? Which pharmacopoeia standards apply? | GMP + ISO 9001, C+A cleanroom |
| Production capacity | Can the supplier meet volume demands without lead time risk? | 3 billion aluminium caps/year |
| Customisation capability | Can dimensions, colour, and stopper type be specified? | Yes — full specification flexibility |
| Quality control depth | Is QC performed in-process or only at final inspection? | Full-process quality control |
| Technical support | Does the supplier offer technical assistance for line integration or qualification? | Professional technical team available |
| Track record | How long has the supplier served the pharmaceutical packaging sector? | Established since 2000 — 20+ years |
Storage, Handling, and Maintenance Guidelines
Proper storage and handling of aluminium caps for infusion bottles is essential to preserve their sealing performance and cleanliness prior to use on the filling line.
Storage Conditions
- Store in a clean, dry environment with controlled temperature (typically 10–30°C) and relative humidity below 65%.
- Avoid direct sunlight exposure, which can degrade rubber stopper elasticity and accelerate oxidation of aluminium surfaces.
- Keep away from chemical storage areas to prevent reactive vapour exposure.
- Maintain original packaging integrity until use; bulk bags and inner liners protect caps from particulate contamination.
Pre-Use Handling
- Introduce caps into the cleanroom according to documented material transfer procedures to prevent particulate and microbial contamination from entering controlled areas.
- Inspect a representative sample from each batch for dimensional conformance, surface defects, and stopper integrity before committing to production runs.
- If pre-washing or silicone coating of stoppers is required by the formulation specification, verify that the cap assembly is compatible with the intended washing protocol.
Shelf Life
When stored correctly under recommended conditions, aluminium caps for infusion bottles typically maintain their physical and functional properties for up to 2–3 years from the date of manufacture. Actual shelf life should be confirmed per the supplier's certificate of conformance and specific formulation compatibility data.
Frequently Asked Questions (FAQ)
Q1: What is the standard diameter of aluminium caps for infusion bottles?
The most common diameters for aluminium caps used on infusion bottles are 20 mm, 28 mm, and 32 mm, corresponding to the bottle mouth finish sizes used for standard small and large-volume parenteral glass containers. Custom diameters can be produced to match specific bottle specifications. Always confirm the cap diameter and bottle finish dimensions with both the cap supplier and bottle manufacturer before finalising the closure system.
Q2: Can aluminium caps for infusion bottles be supplied pre-sterilised?
Most aluminium caps for infusion bottles are supplied clean but not sterile, as terminal sterilisation of the filled container (autoclave) is the standard practice in LVP manufacturing. However, for aseptic filling processes, caps can be provided gamma-irradiated or ethylene oxide (EtO)-treated upon request, subject to compatibility testing. Changjiang Lids produces caps in a C+A cleanroom environment, providing a low-bioburden baseline that supports downstream sterilisation processes.
Q3: What is the difference between bromobutyl and chlorobutyl rubber stoppers in infusion caps?
Both bromobutyl and chlorobutyl are halogenated butyl rubber compounds used for pharmaceutical stopper manufacture. Bromobutyl generally offers superior moisture vapour transmission resistance and is preferred for moisture-sensitive or lyophilised products. Chlorobutyl tends to have slightly better chemical resistance to certain aqueous drug formulations and may be preferred for aqueous injections. The choice should be guided by extractables and compatibility testing with the specific drug product. Suppliers and formulators should refer to USP <381>, EP 3.2.9, or ChP standards for stopper testing requirements.
Q4: How do I verify container closure integrity (CCI) after capping with aluminium infusion caps?
CCI testing for capped infusion bottles can be performed using several methods: vacuum decay (non-destructive, suitable for aqueous-filled containers), helium leak detection (high sensitivity for dry products), dye ingress testing (destructive, used for validation), and high voltage leak detection (HVLD) for aqueous products. The appropriate method depends on the container-closure system and product type. Regulators increasingly favour deterministic (non-dye-based) methods per USP <1207> and corresponding PhEur guidance.
Q5: Are aluminium caps for infusion bottles compatible with automated high-speed capping lines?
Yes. Aluminium caps for infusion bottles are designed for compatibility with automated rotary and linear capping equipment used in pharmaceutical production. Dimensional precision — including consistent skirt height, shell diameter, and cap height — is critical for reliable feeding and crimping in high-speed operations. Caps from Changjiang Lids are produced with tight dimensional tolerances to support efficient automated line integration and minimise capping defect rates.
Q6: Can the aluminium cap colour be customised for different product lines?
Yes. Colour coding is a widely used practice in hospital and pharmacy environments to differentiate between medication types, concentrations, or administration routes — reducing the risk of medication errors. Changjiang Lids offers aluminium caps for infusion bottles in a range of specified colours through anodising or coating processes. Custom colour specifications should be confirmed during the product qualification stage to ensure colour consistency across production batches.
Q7: What regulatory documentation is typically provided with aluminium infusion caps?
Standard regulatory documentation for pharmaceutical-grade aluminium caps for infusion bottles includes: Certificate of Analysis (CoA) with dimensional and functional test data, Certificate of Conformance (CoC), material safety data, extractables/leachables data (where available), and production batch traceability records. GMP-certified suppliers such as Changjiang Lids maintain documentation systems that support pharmaceutical manufacturer DMF filings and supplier qualification audits.

 English
English русский
русский