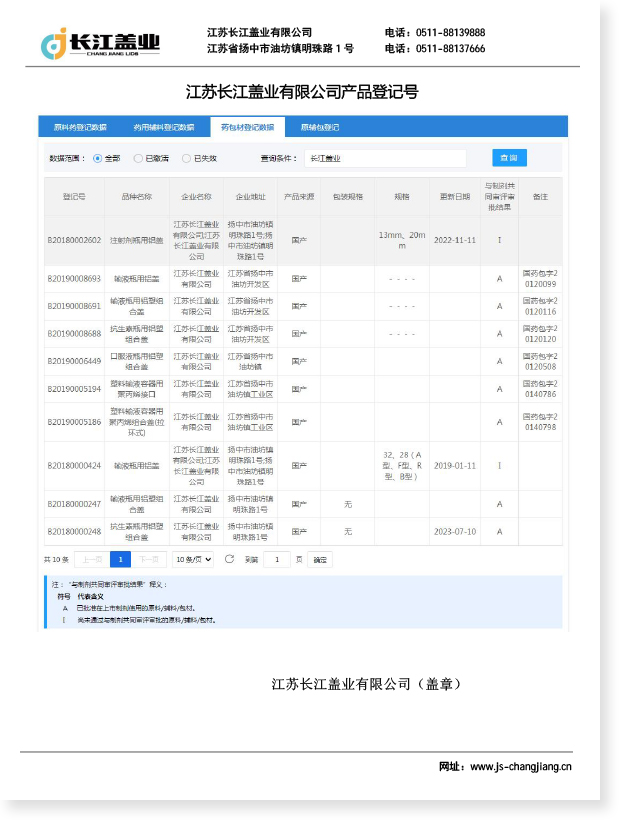
Aluminium caps for injection vials are primary pharmaceutical packaging components designed to seal and secure rubber stoppers on glass or plastic vials used for injectable medications. These caps serve as the final barrier between the drug product and the external environment, playing a critical role in maintaining sterility, preventing contamination, and ensuring tamper evidence throughout the product's shelf life.
Manufactured from high-purity aluminium alloy, aluminium caps for injection vials are widely used in vaccine packaging, antibiotic formulations, lyophilized (freeze-dried) products, and other parenteral drug applications. Their crimp-seal design locks the stopper firmly in place, while the flip-off or tear-off tab enables convenient, aseptic access for healthcare professionals. At Changjiang Lids, each cap is produced under GMP-compliant cleanroom conditions to meet the stringent requirements of global pharmaceutical markets.
The structural integrity and material consistency of aluminium caps for injection vials directly impact drug safety. As such, key performance parameters include dimensional accuracy, sealing force, corrosion resistance, and cleanliness level — all of which must comply with pharmacopoeial standards such as USP, EP, and CP.
Product Structure & Working Principle
A standard aluminium cap for injection vials consists of two primary parts: the aluminium shell and the plastic (usually PP or PE) flip-off disc. The aluminium shell is crimped over the vial neck and rubber stopper during the filling and sealing process, creating a hermetic closure. The plastic disc covers the injection port and can be flipped open without removing the entire cap — allowing needle penetration without compromising the sterile seal.
Sealing Mechanism
During production, the aluminium blank is stamped into a cup-shaped cap, then combined with the plastic button. During vial filling, automated capping machines crimp the aluminium skirt under the vial's flange, compressing the rubber stopper and creating an airtight, tamper-evident seal. The crimping force must be precisely controlled — too low risks leakage; too high can deform the glass vial neck.
Material Composition
Changjiang Lids uses pharmaceutical-grade aluminium alloy (typically 8011 or 1060 series) that meets RoHS and REACH compliance requirements. The plastic disc is made from food-grade polypropylene (PP), available in a range of colors for product differentiation and identification purposes.
Product Specifications Overview
| Cap Diameter | Vial Size (mL) | Common Application | Aluminium Thickness (mm) | Cap Type |
|---|---|---|---|---|
| 13 mm | 2 – 10 mL | Antibiotics, small-volume injectables | 0.20 – 0.25 | Flip-off / Plain |
| 20 mm | 10 – 50 mL | Vaccines, lyophilized products | 0.25 – 0.30 | Flip-off / Tear-off |
| 28 mm | 50 – 100 mL | Large-volume injectables | 0.28 – 0.35 | Flip-off / Crimp-only |
| 32 mm | 100 mL+ | Infusion, high-volume products | 0.30 – 0.40 | Flip-off / Plain |
Changjiang Lids offers customizable cap dimensions and plastic button colors to accommodate different vial formats and branding requirements. Custom printing on the aluminium cap surface is also available for product identification.
Key Performance Parameters
The performance of aluminium caps for injection vials is evaluated across multiple dimensions. Below are the critical parameters and their industry benchmarks:
| Parameter | Requirement | Test Method | Significance |
|---|---|---|---|
| Dimensional Accuracy | ±0.1 mm tolerance | Caliper / CMM | Ensures proper fit on vial neck |
| Seal Integrity | No leakage under pressure | Vacuum / Dye penetration | Prevents contamination, maintains sterility |
| Aluminium Purity | ≥99.0% (8011/1060 alloy) | Spectroscopic analysis | Corrosion resistance, chemical inertness |
| Particulate Cleanliness | ≤1 visible particle / cap | Visual / Light obscuration | Critical for injectable drug safety |
| Flip-off Force | 10 – 30 N (user-friendly range) | Force gauge test | Ease of use in clinical settings |
| Tamper Evidence | Visible deformation on removal | Visual inspection post-removal | Drug integrity and patient safety |
Product Type Comparison: Flip-Off vs. Tear-Off vs. Plain Aluminium Caps
Different pharmaceutical applications call for different cap configurations. The three most common types are compared below:
| Feature | Flip-Off Cap | Tear-Off Cap | Plain Aluminium Cap |
|---|---|---|---|
| Access Method | Flip plastic disc to expose stopper | Tear aluminium tab to remove top | Requires decapping tool |
| Sterile Seal Maintained | Yes (stopper intact) | Yes (stopper intact) | Yes |
| Ease of Use | High – one-hand operation | Medium – requires pulling force | Low – requires tools |
| Common Applications | Vaccines, antibiotics, injectables | Lyophilized drugs, biologics | Veterinary, industrial applications |
| Color Customization | Yes (PP disc color) | Limited | Lacquer coating available |
| Tamper Evidence | High | High | Medium |
Application Cases
Aluminium caps for injection vials are used across a broad range of pharmaceutical segments. The following cases illustrate typical applications:
1. Vaccine Packaging
Multi-dose vaccine vials (e.g., 10-dose influenza or COVID-19 vaccines) rely on 20 mm flip-off aluminium caps to maintain sterility between doses. The crimp seal prevents micro-leakage during temperature cycling in cold-chain logistics. Changjiang Lids' 20 mm caps are used by multiple vaccine manufacturers, meeting WHO prequalification packaging guidelines.
2. Antibiotic Vials
Powder-for-injection antibiotics (e.g., cephalosporins, penicillin) packaged in 5–20 mL vials typically use 13 mm or 20 mm flip-off caps. These caps must withstand the freeze-drying (lyophilization) process without dimensional deformation or seal compromise.
3. Biologic & Protein Therapeutics
High-value biologics demand the highest cleanliness standards. Changjiang Lids' C+A cleanroom-produced caps minimize particulate and endotoxin risk, supporting the packaging of monoclonal antibodies, hormones, and enzyme replacement therapies.
4. Veterinary Injectables
Aluminium caps for injection vials are also widely used in veterinary medicine for vaccines, antiparasitics, and hormonal products packaged in multi-dose glass vials. Plain aluminium caps are commonly preferred for cost-effective high-volume veterinary packaging.
Quality Assurance & Regulatory Compliance
Pharmaceutical packaging components are subject to strict regulatory oversight. Aluminium caps for injection vials must comply with pharmacopoeial monographs and GMP manufacturing standards. Jiangsu Changjiang Lids Co., Ltd. has built its entire quality system around this requirement.
- GMP Compliance: All production lines operate in accordance with national GMP regulations for pharmaceutical packaging.
- ISO 9001 Certified: Quality management from raw material procurement to finished product dispatch is governed by ISO 9001 protocols.
- C+A Level Cleanroom: Changjiang Lids has purpose-built C+A grade cleanroom facilities to ensure ultra-low particle counts during final packaging stages.
- In-Process & Final Testing: Every batch undergoes dimensional inspection, seal integrity testing, visual cleanliness verification, and material traceability review.
- Annual Production Capacity: Over 3 billion aluminium and aluminium-plastic caps per year, supporting reliable global supply chains.
Selection Guide: How to Choose the Right Aluminium Cap
Selecting the appropriate aluminium cap for injection vials requires consideration of multiple technical and regulatory factors. Use the guide below as a starting reference:
- Vial size and neck diameter: Match cap diameter (13 mm, 20 mm, 28 mm, or 32 mm) to the corresponding vial ISO standard.
- Drug type and administration route: High-sensitivity biologics require higher cleanliness-grade caps; standard injectables may use standard-grade products.
- Capping equipment compatibility: Verify that cap dimensions and aluminium hardness are compatible with your capping machine's crimping head specifications.
- Regulatory market: Confirm that cap specifications comply with the pharmacopoeial requirements of your target market (USP, EP, CP, JP).
- Color and branding: Choose PP disc colors or surface printing for product differentiation and medication safety (color-coded dosages).
- Volume and logistics: For large-volume procurement, confirm supplier annual capacity and cleanroom packaging options. Changjiang Lids' production capacity of 3 billion units per year supports both high-volume and specialty orders.
Maintenance & Storage Guidelines for Aluminium Caps
Proper handling and storage of aluminium caps for injection vials is essential to preserve their cleanliness and performance before use:
- Store in a dry, dust-free environment at 15–30°C, away from direct sunlight and chemical vapors.
- Keep caps in their original sealed packaging until use to prevent particulate contamination.
- Follow a FIFO (First In, First Out) inventory system; do not exceed the stated shelf life (typically 24–36 months from production date).
- When introducing caps into cleanroom filling lines, follow your facility's material transfer and depyrogenation SOPs.
- Inspect each delivery batch against the certificate of analysis (CoA) and perform incoming quality checks before releasing for production use.
Frequently Asked Questions (FAQ)
Q1: What is the difference between an aluminium cap and an aluminium-plastic combination cap?
A plain aluminium cap consists entirely of aluminium and typically requires a decapping tool for access. An aluminium-plastic combination cap (also called a flip-off cap) integrates a polypropylene disc that can be flipped open manually to expose the rubber stopper for needle penetration, without removing the aluminium crimp seal. The combination cap is now the dominant format for injectable pharmaceutical packaging due to its clinical convenience.
Q2: Are aluminium caps for injection vials compatible with all capping machines?
Compatibility depends on cap diameter, aluminium alloy hardness (temper grade), and cap height. Most standard capping machines accept 13 mm, 20 mm, 28 mm, and 32 mm caps, but crimping jaws must be adjusted accordingly. Changjiang Lids can provide dimensional data sheets and sample caps for equipment qualification trials upon request.
Q3: What cleanroom grade are Changjiang Lids' caps produced in?
Jiangsu Changjiang Lids Co., Ltd. operates a C+A level cleanroom (ISO Class 7/8 background with ISO Class 5 critical zones), which meets the GMP requirements for pharmaceutical primary packaging production. This ensures that finished caps have minimal particulate burden before entering the customer's filling line.
Q4: Can the plastic disc color be customized?
Yes. Changjiang Lids offers a wide range of standard PP disc colors (including red, blue, yellow, green, white, orange, and more) as well as custom colors with Pantone matching for OEM projects. Color-coded caps are widely used to differentiate drug strength, product line, or brand identity.
Q5: What pharmacopoeial standards do these caps meet?
Aluminium caps for injection vials from Changjiang Lids are designed and tested in accordance with relevant monographs and standards including the Chinese Pharmacopoeia (CP), European Pharmacopoeia (EP), and United States Pharmacopeia (USP). Customers targeting specific markets can request certificates of conformity for the applicable standard.
Q6: What is the minimum order quantity (MOQ)?
MOQ varies by cap specification and customization requirements. Standard sizes are available at lower MOQs, while custom colors or printed caps may require higher minimum quantities. Please contact Changjiang Lids' sales team for specific MOQ and lead time information based on your project requirements.
Q7: How is the seal integrity of the cap verified after crimping?
Seal integrity is typically verified using container closure integrity testing (CCIT) methods such as vacuum-decay testing, dye ingress testing, or headspace gas analysis. These tests are performed on finished, crimped vials — not on the cap alone. The cap's contribution to seal integrity is validated through dimensional compliance and crimping force studies during packaging development.

 English
English русский
русский