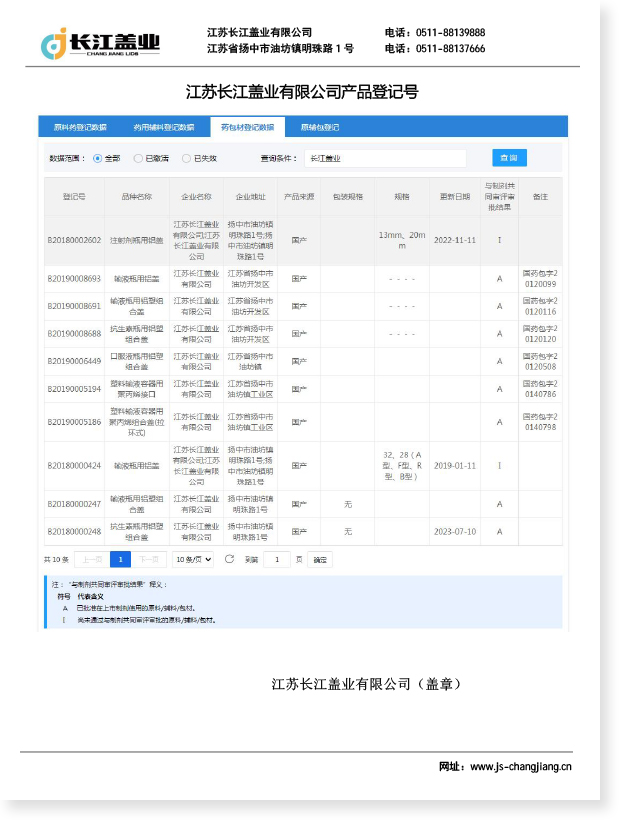
An aluminum-plastic cap for injection vials is a composite primary pharmaceutical packaging component consisting of an outer aluminum shell crimped over an inner plastic (typically polypropylene or polyethylene) insert and a rubber stopper. Together, these elements form a tamper-evident, hermetically sealed closure system that protects injectable drug products — including vaccines, antibiotics, lyophilized formulations, and biologics — from contamination, leakage, and environmental degradation throughout their entire shelf life.
The aluminum component provides mechanical strength and a visible tamper-evidence feature upon removal, while the plastic insert acts as a protective interface between the aluminum and the rubber stopper, preventing direct metal-to-drug contact and reducing particulate risk. As a result, the aluminum-plastic cap for injection vials has become the industry-standard closure solution for parenteral packaging worldwide.
Jiangsu Changjiang Lids Co., Ltd. has been manufacturing pharmaceutical-grade aluminum-plastic caps since 2000, with an annual production capacity of 3 billion aluminum and aluminum-plastic caps, all produced in GMP-compliant C+A level cleanrooms.
Product Structure & Working Principle
Understanding the layered construction of an aluminum-plastic cap helps clarify its sealing performance and compatibility requirements:
Three-Layer Composite Structure
- Outer Aluminum Shell — Typically made from 8011 or 1060 aluminum alloy, the shell is crimped mechanically onto the vial neck. The aluminum provides rigidity, tamper evidence (the flip-off tab tears away on first opening), and a professional appearance suitable for label printing or embossing.
- Inner Plastic Insert (Button/Disc) — Molded from pharmaceutical-grade polypropylene (PP) or low-density polyethylene (LDPE), the plastic insert sits within the aluminum shell. It cushions the rubber stopper, prevents direct aluminum-to-stopper abrasion, and enables the flip-off or tear-off opening mechanism without exposing the stopper to sharp metal edges.
- Rubber Stopper Interface — Although the rubber stopper is a separate component, the aluminum-plastic cap is designed to work in precise dimensional tolerance with standardized stoppers (e.g., ISO 8362-2). The cap assembly maintains compression on the stopper to preserve hermetic integrity.
Sealing Mechanism
During filling and stoppering on a pharmaceutical production line, the rubber stopper is inserted into the vial neck. The aluminum-plastic cap assembly is then placed over the stopper and mechanically crimped using a capping machine. The crimping deforms the aluminum skirt beneath the vial neck flange, locking the assembly in place. The combination of stopper compression and crimped aluminum creates a hermetic seal that prevents microbial ingress, gas exchange, and moisture penetration.
Key Performance Parameters
Pharmaceutical customers evaluate aluminum-plastic caps against a defined set of functional and regulatory performance criteria. The table below summarizes the most critical parameters:
| Parameter | Typical Requirement | Test Method | Significance |
|---|---|---|---|
| Sealing Integrity | No leakage at ≥0.06 MPa vacuum | YBB 00122003 / USP <1207> | Prevents contamination and preserves sterility |
| Dimensional Tolerance | ±0.2 mm on critical dimensions | ISO 8362-3 / GB/T 12255 | Ensures compatibility with standard vials and capping machines |
| Crimp Force Resistance | ≥50 N pull-off force after crimping | Internal tensile test | Tamper evidence and transport integrity |
| Particulate Contamination | ≤20 particles ≥10 µm per cap | Light obscuration / microscopy | Critical for injectable drug safety |
| Heavy Metal Content | Below ICH Q3D limits | ICP-MS analysis | Regulatory compliance for patient safety |
| Sterility / Bioburden | Meets pharmacopeial requirements | USP <71> / EP 2.6.1 | Required for direct contact packaging |
| Flip-Off Tab Removal Force | 15–35 N | Force gauge test | User safety and tamper-evidence balance |
Product Variants & Specifications
Aluminum-plastic caps for injection vials are available in a range of standard sizes and opening configurations to match different vial neck diameters and clinical use scenarios. CHANGJIANG LIDS supplies the full spectrum of standard variants as well as custom specifications.
| Cap Outer Diameter | Nominal Vial Neck (mm) | Opening Type | Typical Application |
|---|---|---|---|
| 13 mm | 13 | Flip-off / Tear-off | Small-volume parenterals, antibiotics |
| 20 mm | 20 | Flip-off / Tear-off | Vaccines, lyophilized products, biologics |
| 28 mm | 28 | Flip-off / Tear-off | Larger-volume injectables, powder vials |
| 32 mm | 32 | Tear-off | High-volume injectables, hospital pharmacy |
Opening Configuration Comparison
| Feature | Flip-Off Cap | Tear-Off Cap |
|---|---|---|
| Opening Method | Plastic button flipped up to expose stopper | Aluminum tab torn away completely |
| Stopper Access | Needle puncture through exposed stopper | Stopper fully accessible after tear-off |
| Tamper Evidence | High — flip tab cannot be resealed | High — tear cannot be reversed |
| Common Use | Vaccines, multi-dose vials, biologics | Single-use injectables, hospital use |
| Capping Machine Compatibility | Standard rotary/linear crimpers | Standard rotary/linear crimpers |
| Color Coding | Available (plastic button) | Limited (aluminum surface) |
Application Scenarios
The aluminum-plastic cap for injection vials serves a wide variety of parenteral drug product segments. Below are representative application examples across therapeutic categories:
Vaccines
Vaccine vials — including multi-dose presentations for influenza, COVID-19, hepatitis B, and other immunizations — rely on aluminum-plastic caps to maintain sterility across multiple needle insertions. The flip-off configuration allows repeated puncture while the crimped aluminum skirt maintains seal integrity and provides visible evidence if the vial has been tampered with.
Antibiotic Powder for Injection
Lyophilized or crystalline antibiotic powders (e.g., penicillin, cephalosporins) require hermetic closures that prevent moisture ingress — a key cause of product degradation. The aluminum-plastic cap provides a moisture barrier, with the plastic insert preventing direct aluminum contact with moisture-sensitive formulations.
Biologics & Lyophilized Products
Biologics such as monoclonal antibodies and lyophilized proteins require packaging with minimal extractables and leachables. CHANGJIANG LIDS manufactures caps using pharmaceutical-grade materials that comply with ICH Q3D elemental impurity guidelines, supporting compatibility studies and regulatory dossier submissions.
Small-Volume Parenterals (SVP)
Vials for anesthetics, analgesics, contrast agents, and other SVPs with volumes from 1 mL to 50 mL are sealed with 13 mm or 20 mm aluminum-plastic caps. High-speed automated filling lines require caps with consistent dimensional tolerances to ensure reliable capping machine throughput exceeding 30,000 vials/hour.
Aluminum-Plastic Cap vs. Pure Aluminum Cap: Detailed Comparison
Pharmaceutical manufacturers choosing between aluminum-plastic caps and conventional all-aluminum caps should consider the following performance and operational differences:
| Comparison Dimension | Aluminum-Plastic Cap | Pure Aluminum Cap |
|---|---|---|
| Stopper Protection | High — plastic insert cushions stopper | Moderate — direct metal-to-stopper contact |
| Particulate Risk | Lower — plastic prevents aluminum abrasion | Higher — metal-to-rubber abrasion possible |
| Opening Convenience | Flip-off plastic button — fingertip operation | Requires cap opener tool in some designs |
| Color Coding Options | Wide — plastic button available in many colors | Limited — surface lacquer only |
| Regulatory Trend | Preferred by modern pharmacopeias for injectables | Still used for some oral liquid/infusion formats |
| Compatibility with Lyophilization | Excellent — designed for lyophilized vials | Less optimized |
| Unit Cost | Slightly higher (composite material) | Lower |
Regulatory Standards & Compliance
Aluminum-plastic caps for injection vials are subject to rigorous national and international pharmaceutical packaging standards. CHANGJIANG LIDS designs and manufactures all products to comply with the following regulatory frameworks:
- China National Standard: YBB 00122003 (Aluminum Cap for Pharmaceutical Use), GB/T 12255
- International Standard: ISO 8362-3 (Aluminium caps for injection vials)
- European Pharmacopoeia (EP): Ph. Eur. 3.2.5 (Tamper-evident packaging for injectable products)
- US Pharmacopeia (USP): USP <660> Containers — Glass, USP <1207> Package Integrity Evaluation
- GMP Compliance: Manufactured in C+A cleanroom per China GMP and ICH Q7 guidelines
- Quality System: ISO 9001:2015 certified quality management system
CHANGJIANG LIDS maintains complete traceability documentation for raw materials, in-process quality data, and finished product release testing, supporting customer regulatory submissions including Drug Master Files (DMF) and CEP applications.
Quality Inspection & Testing Protocol
Every batch of aluminum-plastic caps undergoes a multi-stage quality inspection program before shipment. The following table outlines the standard testing protocol:
| Inspection Stage | Test Item | Acceptance Criterion | Frequency |
|---|---|---|---|
| Incoming Raw Material | Aluminum alloy grade, PP resin certification | CoA match; no heavy metals above limits | Every lot |
| In-Process | Dimensions, visual defects, assembly fit | Per AQL 1.0 sampling plan | Every production shift |
| Finished Product | Seal integrity, flip-off force, appearance | 100% visual; statistical seal test | Every batch |
| Periodic Testing | Extractables, bioburden, particulates | Per pharmacopeial limits | Quarterly / per customer agreement |
Storage, Handling & Maintenance Guidelines
Proper storage and handling of aluminum-plastic caps prior to use on the filling line is essential to maintaining product quality and regulatory compliance.
Storage Conditions
- Store in original sealed packaging at 15–30°C, relative humidity ≤60%.
- Keep away from direct sunlight and sources of chemical contamination.
- Shelf life is typically 24 months from manufacture date when stored correctly.
- Store in a dedicated pharmaceutical packaging warehouse, separated from non-pharmaceutical materials.
Pre-Use Handling
- Transfer caps from storage to the cleanroom environment through appropriate airlocks and depyrogenation procedures if required.
- Inspect for transport damage (dents, loose plastic inserts, discoloration) before loading into capping hoppers.
- Do not open packaging until caps are ready for use; once opened, use within the period specified in your validated process.
Capping Machine Maintenance
- Verify crimping head dimensions match cap size at each product changeover.
- Monitor crimp quality (residual seal force, cap tilt angle) at defined intervals during production runs.
- Schedule preventive maintenance per equipment manufacturer guidelines to prevent seal integrity failures.
Frequently Asked Questions (FAQ)
Q1: What is the difference between an aluminum-plastic cap and a plain aluminum cap for injection vials?
An aluminum-plastic cap incorporates a molded plastic (typically polypropylene) insert inside the aluminum shell. This insert cushions the rubber stopper, reduces the risk of aluminum particulates from metal-to-rubber abrasion, and enables a convenient flip-off opening mechanism. A plain aluminum cap lacks the plastic insert, resulting in direct metal-to-stopper contact. For modern injectable drug packaging — particularly vaccines and biologics — the aluminum-plastic cap is the preferred format due to its superior particulate performance and user convenience.
Q2: What sizes of aluminum-plastic caps does CHANGJIANG LIDS supply?
CHANGJIANG LIDS supplies aluminum-plastic caps in standard diameters of 13 mm, 20 mm, 28 mm, and 32 mm, corresponding to the most widely used injection vial neck finishes per ISO 8362 and Chinese pharmacopeial standards. Custom sizes and colors are available on request, with tooling support from our in-house technical team.
Q3: Are the caps compatible with standard pharmaceutical filling and capping lines?
Yes. CHANGJIANG LIDS aluminum-plastic caps are manufactured to ISO 8362-3 dimensional tolerances, ensuring compatibility with rotary and linear capping equipment from major manufacturers including Bausch+Ströbel, IMA, Groninger, and others. All critical dimensions are controlled within ±0.2 mm to support high-speed automated capping at rates exceeding 30,000 vials/hour.
Q4: What cleanroom grade are the caps manufactured in?
All aluminum-plastic caps for injection vials at CHANGJIANG LIDS are produced in C+A level cleanrooms, which correspond to ISO Class 7 background with ISO Class 5 critical zones. This exceeds the minimum requirements for primary pharmaceutical packaging and ensures low bioburden and particulate levels upon delivery.
Q5: Can aluminum-plastic caps be color-coded for product differentiation?
Yes. The plastic insert (flip-off button) can be produced in a wide range of colors using pharmaceutical-grade masterbatches that comply with regulatory requirements for direct contact packaging. Color coding is commonly used by pharmaceutical companies to differentiate drug concentrations, vial volumes, or product lines at the point of care, reducing medication errors. CHANGJIANG LIDS supports custom color matching and can provide color consistency data across production batches.
Q6: What documentation is available to support regulatory submissions?
CHANGJIANG LIDS provides a comprehensive documentation package including: Certificate of Analysis (CoA), Certificate of Conformance (CoC), material safety data (raw material CoAs and heavy metal test reports), dimensional drawings, and extractables/leachables data. Customers requiring Drug Master File (DMF) support or CEP assistance can contact our technical team. Our ISO 9001:2015 certification and GMP compliance records are available for customer audits.
Q7: What is the minimum order quantity (MOQ) and lead time?
Standard MOQs and lead times vary by cap size, color, and customization requirements. With an annual production capacity of 3 billion aluminum and aluminum-plastic caps, CHANGJIANG LIDS is equipped to fulfill both high-volume commercial orders and smaller validation batches. Contact our sales team for specific pricing and delivery schedules tailored to your project requirements.
Q8: How should I troubleshoot if caps are causing capping machine jams?
Machine jams are most commonly caused by dimensional variation exceeding equipment tolerances, improper hopper orientation, or contaminated/deformed caps. Recommended troubleshooting steps include: (1) verify cap lot dimensions against the equipment's qualification specifications; (2) inspect the hopper and chute for debris; (3) check capping head wear and alignment; (4) review storage conditions to rule out cap deformation from humidity or temperature. CHANGJIANG LIDS' technical support team can assist with root cause analysis and provide dimensional data from the relevant production lot.

 English
English русский
русский