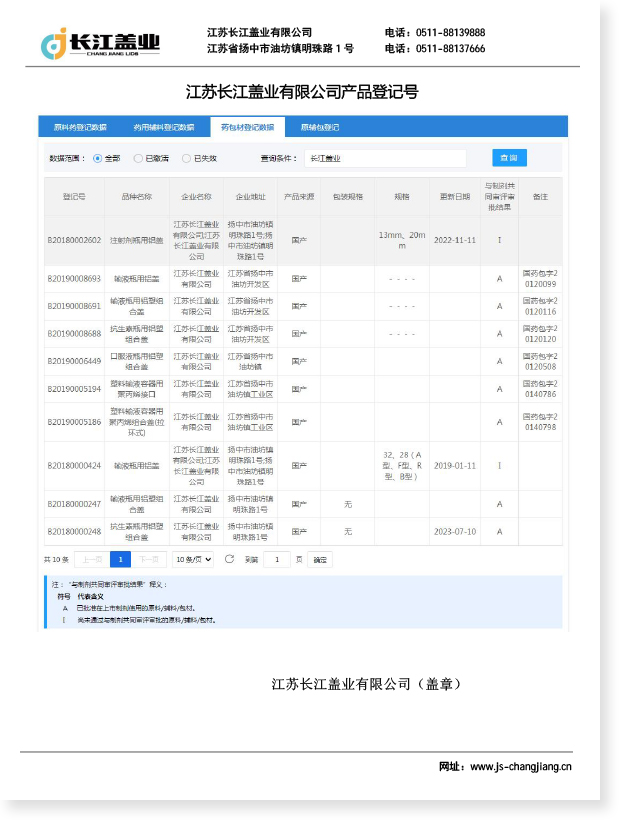
Euro PP Caps are high-performance polypropylene pharmaceutical packaging closures independently developed and produced by Jiangsu Changjiang Lids Co., Ltd., adhering to pharmaceutical industry standards. Euro PP Caps adopt food-grade polypropylene raw materials, matching the sealing and protection requirements of pharmaceutical infusion containers and medical packaging.
Euro PP Caps feature stable physical and chemical properties, excellent sealing performance and corrosion resistance, effectively isolating external pollutants and ensuring the safety and stability of pharmaceutical preparations. Changjiang Lids produces Euro PP Caps in a C+A level cleanroom, fully complying with GMP regulations and ISO 9001 quality system, with an annual production capacity of 800 million pieces, providing standardized and high-quality packaging solutions for global pharmaceutical enterprises.
Working Principle of Euro PP Caps
Euro PP Caps rely on the structural design of polypropylene materials and precise mold matching to achieve physical sealing. The product forms a tight fit with the bottle mouth through compression and clamping, blocking gas, moisture and microorganisms. Changjiang Lids optimizes the structural parameters of Euro PP Caps through professional R&D, ensuring consistent sealing effect and easy operation in pharmaceutical production lines.
Application Cases of Euro PP Caps
- Applied to infusion container sealing, matching large-scale infusion pharmaceutical production lines of domestic and foreign customers
- Used for oral liquid and injection packaging, providing sterile protection for pharmaceutical preparations
- Suitable for medical disposable packaging systems, meeting international pharmaceutical packaging transportation and storage requirements
- Cooperated with pharmaceutical manufacturers in Asia, Europe and North America, Euro PP Caps have been verified in long-term clinical and commercial applications
Performance Comparison of Euro PP Caps and Similar Products
| Performance Index | Euro PP Caps | Aluminum-plastic Caps | Aluminum Caps |
|---|---|---|---|
| Material | Food-grade Polypropylene | Aluminum + Plastic | Pure Aluminum |
| Sealing Performance | Excellent | Good | Medium |
| Chemical Stability | High | Medium | Low |
| Production Environment | C+A Cleanroom | General Cleanroom | General Workshop |
| Applicable Scenarios | Infusion/Injection/Oral Liquid | Antibiotic/Vaccine | Conventional Injection |
Maintenance and Storage Guidelines for Euro PP Caps
Storage Conditions
Store Euro PP Caps in a dry, ventilated and pollution-free environment, avoid direct sunlight and high temperature, and the storage temperature is controlled at 10℃-30℃.
Handling Requirements
Handle with care during transportation and use to avoid extrusion and deformation; strictly implement aseptic operation standards to prevent secondary pollution.
Quality Inspection
Changjiang Lids provides full-process quality control for Euro PP Caps, and users can conduct appearance and sealing tests before use to ensure product performance meets standards.
Frequently Asked Questions about Euro PP Caps
- Q: Are Euro PP Caps produced in a cleanroom?
A: Yes. Euro PP Caps are produced in Changjiang Lids’ C+A level cleanroom, fully complying with pharmaceutical GMP regulations. - Q: What is the service life of Euro PP Caps?
A: Under standard storage conditions, the shelf life of Euro PP Caps is 3 years, suitable for long-term pharmaceutical packaging matching. - Q: Can Euro PP Caps be customized?
A: Changjiang Lids supports customized specifications and sizes of Euro PP Caps according to customer pharmaceutical packaging needs, with professional technical team supporting design and production. - Q: Do Euro PP Caps meet international quality certification?
A: Euro PP Caps produced by Changjiang Lids comply with ISO 9001 quality system and pharmaceutical industry GMP standards, meeting international pharmaceutical packaging import and use requirements. - Q: What is the production capacity of Euro PP Caps?
A: Changjiang Lids has an annual production capacity of 800 million pieces for Euro PP Caps, which can meet the large-scale procurement needs of pharmaceutical enterprises.

 English
English русский
русский