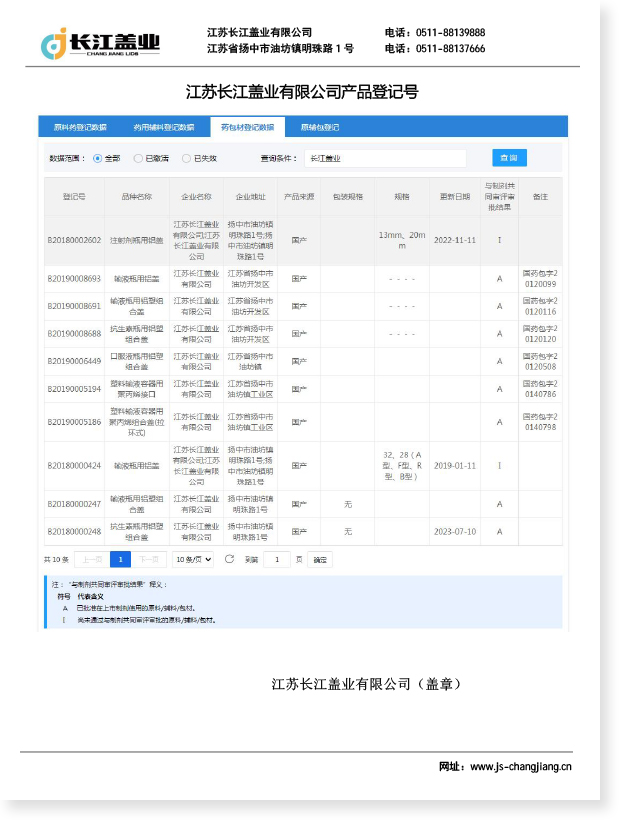
The Aluminum-Plastic Cap for Infusion Bottles is a primary pharmaceutical packaging component designed to seal large-volume parenteral (LVP) infusion containers, including glass and plastic infusion bottles ranging from 50 mL to 1000 mL. Combining an aluminum shell with an integrated plastic (typically polypropylene) flip-off button, these caps create a tamper-evident, sterile closure that meets strict regulatory and clinical requirements.
The core functions of an aluminum-plastic cap for infusion bottles include sealing integrity to prevent contamination, compatibility with standard rubber stoppers, and ease of access during clinical administration via the flip-off design. The cap's aluminum shell is crimped tightly around the bottle neck, while the plastic center tab can be flipped open to expose the stopper for needle insertion or infusion set connection — without removing the entire closure.
At Jiangsu Changjiang Lids Co., Ltd., we have specialized in pharmaceutical packaging since 2000. Our aluminum-plastic caps for infusion bottles are manufactured in a C+A level cleanroom under GMP and ISO 9001 quality systems, with an annual output capacity of up to 3 billion aluminum and aluminum-plastic caps — ensuring consistent supply and quality assurance for pharmaceutical manufacturers worldwide.
Product Structure & Working Principle
Understanding the structure of an aluminum-plastic infusion cap helps evaluate its performance in sterile sealing and clinical use.
Key Components
- Aluminum shell: Formed from pharmaceutical-grade aluminum foil (commonly 8011 alloy), providing structural rigidity and airtight crimping onto the bottle neck.
- Plastic flip-off tab: Injection-molded from medical-grade polypropylene (PP), snapped into the aluminum shell center. The tab flips open to reveal the rubber stopper without disturbing the peripheral seal.
- Rubber stopper interface: The cap is designed to work in combination with a butyl rubber stopper that sits beneath, maintaining the sterile barrier and allowing needle penetration.
Sealing Mechanism
During capping, automated crimping equipment applies uniform radial and axial force, rolling the aluminum skirt under the bottle's neck ring. This mechanical deformation creates a hermetic seal. The flip-off PP button remains integral until a nurse or clinician manually lifts it, exposing the stopper for aseptic access — a tamper-evidence feature required by international pharmacopeial standards such as USP, EP, and ChP.
Material Specifications
| Component | Material | Standard / Grade | Key Property |
|---|---|---|---|
| Aluminum Shell | Aluminum Alloy 8011 / 1060 | YS/T 95, GB/T 3198 | High ductility, uniform crimping |
| Flip-Off Button | Medical-Grade Polypropylene (PP) | USP Class VI / ISO 10993 | Non-toxic, sterilization-compatible |
| Surface Finish | Lacquered / Plain / Color-coded | Pharmaceutical coating standards | Corrosion resistance, brand identification |
| Cleanliness | C+A Cleanroom Produced | GMP / ISO 9001 | Particulate control, sterility assurance |
Common Specifications & Dimensions
Aluminum-plastic infusion caps are manufactured in standardized diameters to fit the most widely used infusion bottle neck finishes globally.
| Cap Diameter (mm) | Bottle Volume Range | Typical Application | Compatible Container |
|---|---|---|---|
| 20 mm | 50 mL – 100 mL | Small-volume infusion | Glass / PET bottle |
| 28 mm | 100 mL – 250 mL | Standard IV solutions | Glass bottle |
| 32 mm | 250 mL – 500 mL | Saline, glucose infusion | Glass bottle |
| 38 mm | 500 mL – 1000 mL | Large-volume parenteral | Glass bottle |
Custom dimensions and color-coded flip tabs are available. Changjiang Lids supports OEM specifications to match individual filling line and bottle neck requirements.
Aluminum-Plastic Cap vs. Pure Aluminum Cap: Key Differences
Pharmaceutical manufacturers often choose between pure aluminum caps and aluminum-plastic combination caps depending on their clinical and regulatory requirements.
| Feature | Aluminum-Plastic Cap | Pure Aluminum Cap |
|---|---|---|
| Tamper Evidence | High — flip-off tab visible | Moderate |
| Clinical Access | Easy flip-open, no tools required | Requires full removal or tear-off |
| Sealing Performance | Excellent (combined metal + polymer) | Excellent |
| Color Coding | Available via PP button color | Limited (surface coating only) |
| Sterility Maintenance | Full stopper protection before use | Full stopper protection before use |
| Regulatory Acceptance | USP, EP, ChP compliant | USP, EP, ChP compliant |
| Material Weight | Slightly heavier | Lighter |
| Typical Use Case | Hospital IV bags, LVP solutions | Antibiotic vials, injections |
Application Scenarios
The aluminum-plastic cap for infusion bottles serves a broad range of pharmaceutical and clinical environments:
- 0.9% Sodium Chloride (Normal Saline): The most common LVP solution. Requires consistent sealing integrity and particulate-free surfaces to meet hospital pharmacy standards.
- 5% / 10% Glucose (Dextrose) Solutions: Used in nutritional support IV therapy. Requires caps compatible with slightly acidic solutions and terminal sterilization (121°C autoclave).
- Compound Sodium Chloride Injection: Electrolyte replacement solutions used in emergency medicine and surgical recovery.
- Total Parenteral Nutrition (TPN): Complex lipid and amino acid solutions requiring superior chemical compatibility and long shelf-life seal performance.
- Antibiotic Infusion Solutions: Reconstituted antibiotics (e.g., cephalosporins, vancomycin) administered via IV drip, requiring precise fit with standard bottle necks and rubber stoppers.
- Ophthalmic Irrigation Solutions: Sterile irrigating fluids used in surgical procedures, demanding the highest particulate cleanliness standards.
Changjiang Lids supplies aluminum-plastic infusion caps to domestic pharmaceutical manufacturers as well as export markets across Asia, Europe, and emerging markets, with products supporting both glass bottle and blow-fill-seal (BFS) container lines.
Quality Standards & Regulatory Compliance
Pharmaceutical packaging components for injectable and infusion products are regulated to the highest standards. Aluminum-plastic caps for infusion bottles must meet requirements in the following key areas:
| Test Parameter | Standard Reference | Acceptance Criteria |
|---|---|---|
| Seal Integrity (Leak Test) | USP ⟨1207⟩ / YBB Standard | No leakage at specified pressure |
| Particulate Contamination | ChP 2020 / EP 2.9.19 | ≤ defined particle counts per container |
| Chemical Resistance | ISO 10993-5 / USP Biological Tests | No cytotoxicity, no extractables above threshold |
| Sterilization Compatibility | 121°C / 30 min Autoclave | No deformation, seal maintained |
| Flip-Off Force | Internal / Customer specification | Consistent removal force within defined range |
| Dimensional Tolerance | Customer drawing / ISO standard | ± 0.1–0.2 mm on critical dimensions |
| Heavy Metals / Extractables | ICH Q3D / EP 3.1.9 | Below permitted daily exposure limits |
Changjiang Lids operates under GMP regulations and ISO 9001 quality system certification, with in-house laboratory testing instruments covering dimensional measurement, seal integrity, cleanliness, and material compatibility. Quality control spans the entire production process — from incoming material inspection to finished product release.
Production Process Overview
The manufacturing of aluminum-plastic infusion caps involves several controlled process steps to ensure consistent quality:
- Aluminum coil slitting: Pharmaceutical-grade aluminum coil is precision-slit to the required width for cap blanks.
- Stamping and forming: Automated presses form the aluminum shell with precise dimensional control.
- Surface treatment: Lacquering or anodizing is applied to protect against oxidation and enable color identification.
- PP button injection molding: Medical-grade PP is injection-molded into flip-off tabs in a controlled environment.
- Assembly: PP buttons are mechanically assembled into the aluminum shells with consistent snap force.
- Cleaning and washing: Assembled caps undergo ultrasonic or spray cleaning to achieve the required particulate cleanliness level.
- Cleanroom packaging: Products are packaged in C+A level cleanrooms to prevent recontamination prior to delivery.
- QC inspection and release: Final batch inspection against dimensional, visual, and functional specifications before shipment.
Storage, Handling & Maintenance Guidelines
Proper handling of aluminum-plastic infusion caps is critical to maintain their quality through to point of use:
Storage Conditions
- Store in a cool, dry, clean environment — recommended temperature: 10–30°C, relative humidity ≤ 65%.
- Avoid direct sunlight, moisture, and chemical vapors that may affect the aluminum surface or PP button properties.
- Keep inner packaging sealed until loading into the capping machine to minimize particulate exposure.
- Observe FIFO (First In, First Out) inventory rotation; typical shelf life is 2 years from manufacturing date when stored properly.
Handling Precautions
- Do not drop, crush, or expose caps to mechanical shock, which can distort the aluminum shell geometry and compromise crimping performance.
- Inspect each production lot for visual defects (scratches, deformed skirts, missing PP buttons) before feeding into automated capping equipment.
- Ensure the capping machine's crimping tooling is regularly calibrated to the specified cap and bottle neck dimensions to prevent under-crimping (leak risk) or over-crimping (stopper damage).
Frequently Asked Questions (FAQ)
Q1: What is the difference between an aluminum-plastic cap and a polypropylene (PP) cap for infusion bottles?
An aluminum-plastic cap combines an aluminum crimping shell with a polypropylene flip-off tab. The aluminum component provides strong mechanical sealing through crimping and is used primarily with glass infusion bottles. A polypropylene cap (also called a PP screw or snap cap) is an all-plastic closure typically used with plastic infusion containers (PE or PP bottles), offering lightweight construction and compatibility with blow-fill-seal or injection-blow-molded bottle systems. Changjiang Lids supplies both types to accommodate different container formats.
Q2: Can aluminum-plastic infusion caps withstand terminal sterilization by autoclave?
Yes. Standard aluminum-plastic caps for infusion bottles are designed and tested for compatibility with terminal steam sterilization at 121°C for 30 minutes. The medical-grade polypropylene button retains its dimensional stability and snap-fit integrity under these conditions. Customers should confirm sterilization parameters with supplier test data, especially for non-standard cycle temperatures or durations.
Q3: How is the cleanroom grade of C+A relevant to infusion cap quality?
Infusion products are classified as sterile injectables, subject to very low particulate limits per ChP, EP, and USP guidelines. A C+A level cleanroom (ISO Class 7 background with ISO Class 5 local protection) is the standard GMP environment required for the washing, assembly, and packaging of primary closures for sterile products. Manufacturing in this environment ensures that particulate counts in finished caps meet pharmacopeial requirements, reducing the risk of contamination during the filling and sealing process.
Q4: What aluminum alloy is used, and is it safe for pharmaceutical use?
Pharmaceutical-grade aluminum caps are made from alloy 8011 or 1060, both approved for pharmaceutical packaging contact applications. These alloys are selected for their high ductility (enabling clean crimping without cracking), low heavy metal content, and corrosion resistance when lacquered. They comply with international pharmacopeial standards and are widely accepted by regulatory authorities globally.
Q5: Can caps be customized with color-coded flip-off buttons for different drug products?
Yes. Color coding is a well-established safety practice in hospital pharmacy to differentiate drug types, concentrations, or administration routes at a glance. The polypropylene flip-off button can be produced in a wide range of colors using pharmaceutical-approved pigments. Changjiang Lids offers color customization as part of its OEM service, with minimum order quantities applicable depending on color and cap size.
Q6: What is the typical shelf life of aluminum-plastic infusion caps?
When stored under recommended conditions (cool, dry, clean environment; sealed inner packaging), aluminum-plastic infusion caps have a shelf life of 2 years from the date of manufacture. Beyond this period, dimensional stability, surface quality, and cleanliness should be reverified before use. Changjiang Lids provides manufacturing date and lot traceability on all product packaging.
Q7: How do I verify that an aluminum-plastic infusion cap will fit my specific bottle?
Compatibility depends on three key parameters: cap diameter, bottle neck outer diameter, and neck finish height. Provide your bottle supplier's technical drawing or a physical bottle sample to Changjiang Lids for fitment verification. Our technical team can evaluate dimensional compatibility, recommend the appropriate cap specification, and arrange sample trials before bulk ordering.
Q8: Does Changjiang Lids provide quality documentation such as COA and test reports?
Yes. Jiangsu Changjiang Lids Co., Ltd. provides a Certificate of Analysis (COA) with every production lot, covering key quality parameters including dimensions, appearance, seal integrity, and cleanliness. Additional test reports (e.g., biocompatibility, extractables, sterilization compatibility) can be provided upon request to support regulatory submission requirements.

 English
English русский
русский